“Trying to prevent clinically relevant cell death“
Ever since Rudolf Virchow indicated necrosis as an uncontrollable clinical burden, the concept of necrosis as a non-regulated process evolved. However, today it is known that necrosis results from the activation of genetically determined signalling pathways, and therefore represents a therapeutic target.
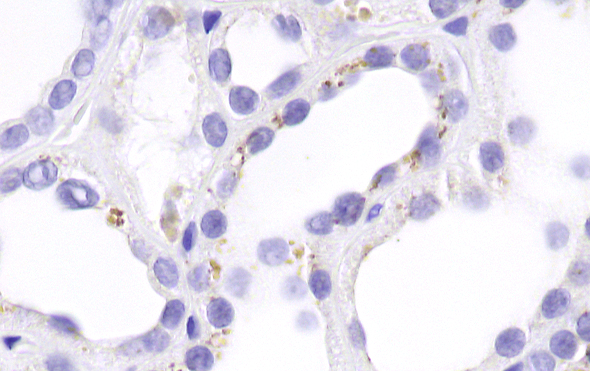
Immunohistochemistry staining of phosphorylated MLKL (activity in the necroptosis pathway) in a human sample of acute kidney injury.
Virchow anticipated that necrosis is a central pathophysiological driver in countless detrimental diseases, such as stroke, myocardial infarction, sepsis, solid tumors, acute hemorrhage, pancreatitis, acute kidney injury, acute liver failure, trauma, rhabdomyolysis and toxicities, just to name the most relevant ones. Therefore, it has been a dream to prevent necrosis pharmacologically in clinical settings. And yes - we belive that this is possible!
Necrosis comes in different flavors, such as kinase-mediated necroptosis, caspase-mediated pyroptosis or lipoxygenase-mediated ferroptosis. Therefore, such enzymes represent novel therapeutic targets. Small molecules may be useful to prevent clinically relevant cell death. However, even larger molecules, such as phenytoin, may carry the potential to prevent clinically relevant necrosis. For 2017 update on ferroptosis published in "Cell" click here. For our current understanding of necroinflammation, find a recent review published in physiological reviews here.